Whether drugs, medical products, or anything else, defective products can create a huge negative impact in one’s life. We’re here to ensure you get the compensation you deserve after falling prey to a defective product.
Mazor X Surgical System
The latest Medtronic urgent field safety notice is frightening. The medical device company is warning doctors, surgical centers and hospitals about its Mazor X Surgical System (SST2) – Positioner Type II.
Inclined Sleeper Lawsuits
The Consumer Product Safety Commission has recalled more than 5.6-million infant inclined sleepers since 2019. The once must-have baby product is responsible for at least 73 infant deaths and 1,000s of injuries due to its defective, unsafe design.
Defective Medical Products
Meyers & Flowers is currently litigating cases on behalf of individuals and families who have been harmed by defective medical products.
Medtronic O-Arm O2 Imaging System
An essential imaging system used by doctors during brain and spinal surgeries has a serious malfunction putting patients at risk for severe and possibly fatal injuries.
Medtronic Recall
An FDA recall has been issued on Medtronic Cardiac Resynchronization Therapy and Implantable Cardioverter Defibrillators due to manufacturing errors in 48 units.
Pam Cooking Spray Lawyer
There have been reported cases of Pam Cooking Spray exploding, causing life-threatening injuries. We are committed to helping those affected.
Pressure Cooker Litigation
Pressure cookers can be a timesaving appliance, but because they cook foods quickly through the production of high heat and pressure.
Product Defects & Liability
Countless people are severely injured or killed as a result of accidents caused by defective products or inadequate product instructions or warnings.
Talcum Powder
Meyers & Flowers is reviewing claims on behalf of women who developed ovarian cancer after using talcum or baby powder near the genitals.
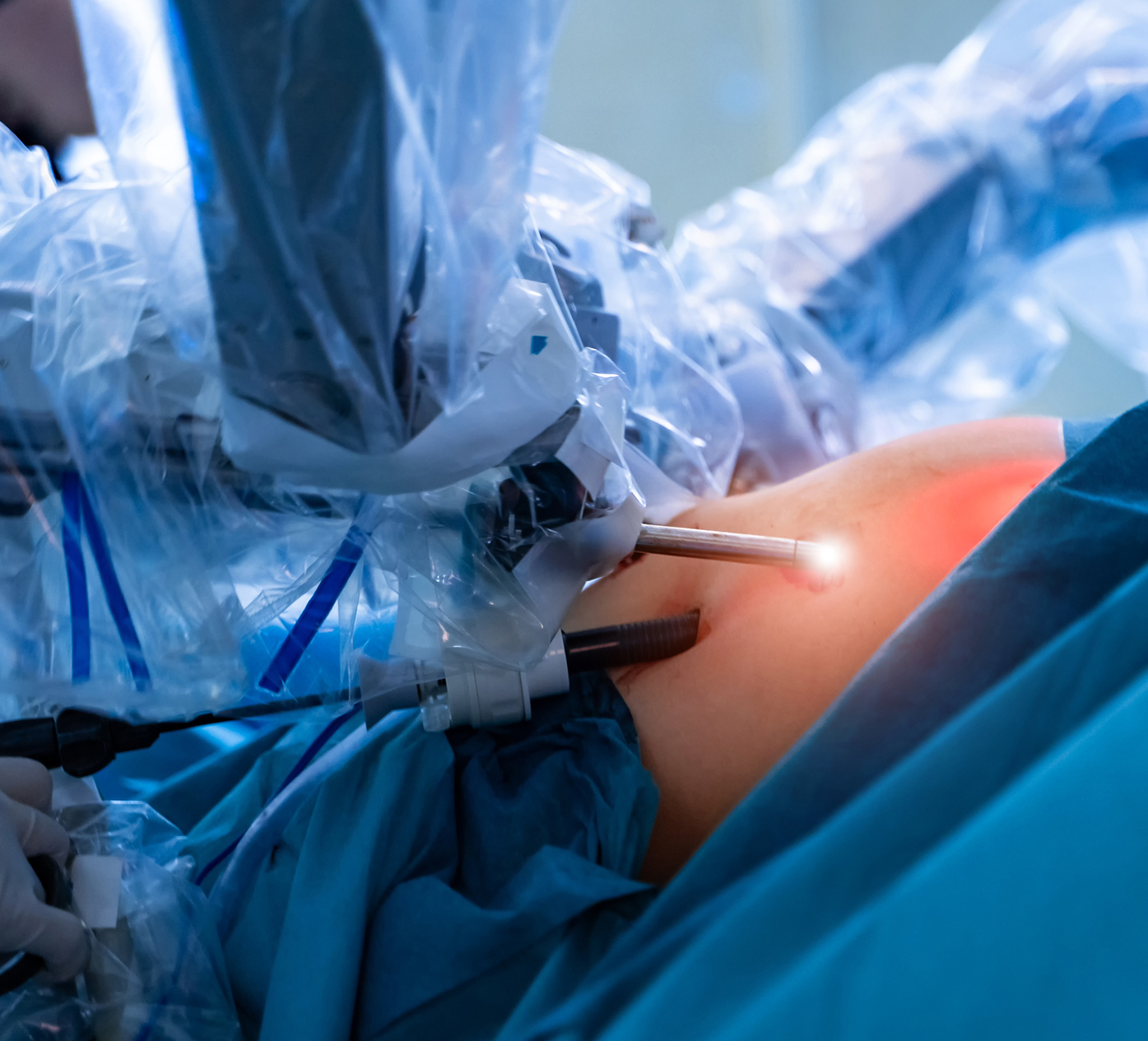
Acist Kodama Intravascular Ultrasound Catheter Recall Litigation
The U.S. Food and Drug Administration has announced a Class I recall of 490 units of the ACIST Kodama Intravascular Ultrasound Catheter due to the high risk of an O-ring within the unit breaking while in use.

Paraquat Herbicide Lawsuits
Recent research has linked paraquat herbicide exposure to Parkinson’s Disease. Meyers & Flowers is investigating and reviewing claims on behalf of farmers and agricultural communities who developed Parkinson’s Disease from the continued use of paraquat for weed and grass control.
EzriCare Artificial Tear Recall
The CDC is investigating a multistate disease outbreak associated with potential bacterial contamination in EzriCare’s lubricant eye drops.